Prior Approval - Other Request
The Other Request type is used in different ways depending on the agency providing the award. Prior Approval "Other Request" can be used for both FDA and NIH awards. The Other Request type appears only for signing officials (SOs) and does not appear for program directors/principal investigators (PD/PIs).
For FDA awards, the Other Request type provides a generic request type for an SO to make requests for FDA grants. All FDA grants are eligible for the Other Request type.
For NIH awards, reach out to your Grants Management Staff (GMS) or Program Officer (PO) for instructions on when to use the Other Request type. NIH awardees should NOT use the Prior Approval "Other Request" type to submit updates to their approved Data Management and Sharing (DMS) Plan. Instead, they should use the DMS Request type to submit an updated DMS Plan; see Prior Approval - DMS Request.
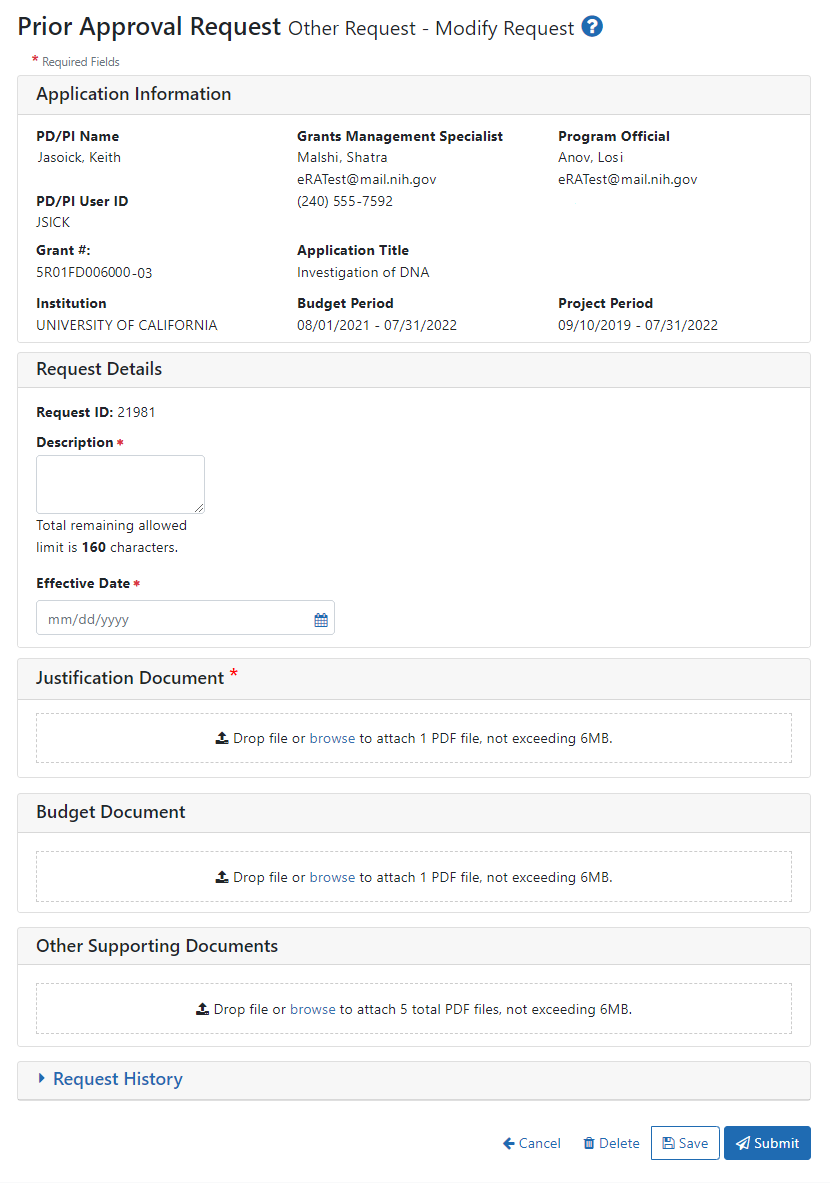
See Viewing and Initiating Requests for details on initiating a request. After initiating an Other Request, you see the following details screen:
-
Must be an FDA or NIH award
-
Must be awarded
-
Must be within the current budget period
-
Grant must not be closed or terminated.
The Other Request can be initiated for any application type matching the above criteria.
-
Fill out all required fields (which have a red asterisk). You can drag and drop a PDF file from your file system onto the Drop file area, or click Browse to locate and select a file.
-
To view or remove a file you have uploaded, click the three-dot ellipsis menu and select View or Delete. If you upload the wrong file, you must delete it in order to upload another, as only one file is allowed to be uploaded.
All uploaded documents must be in PDF format.
