Reviewer is a Federally Registered Lobbyist
A federally registered lobbyist whose term falls within the dates of a review meeting cannot be granted access to the review meeting. A reviewer’s status as a federally registered lobbyist must be indicated on the Confidentiality Agreement screen.
To access and sign the Confidentiality Agreement and indicate that you ARE a federally registered lobbyist:
-
Select the Confidentiality Statement link from the Action column of the List of Meetings.
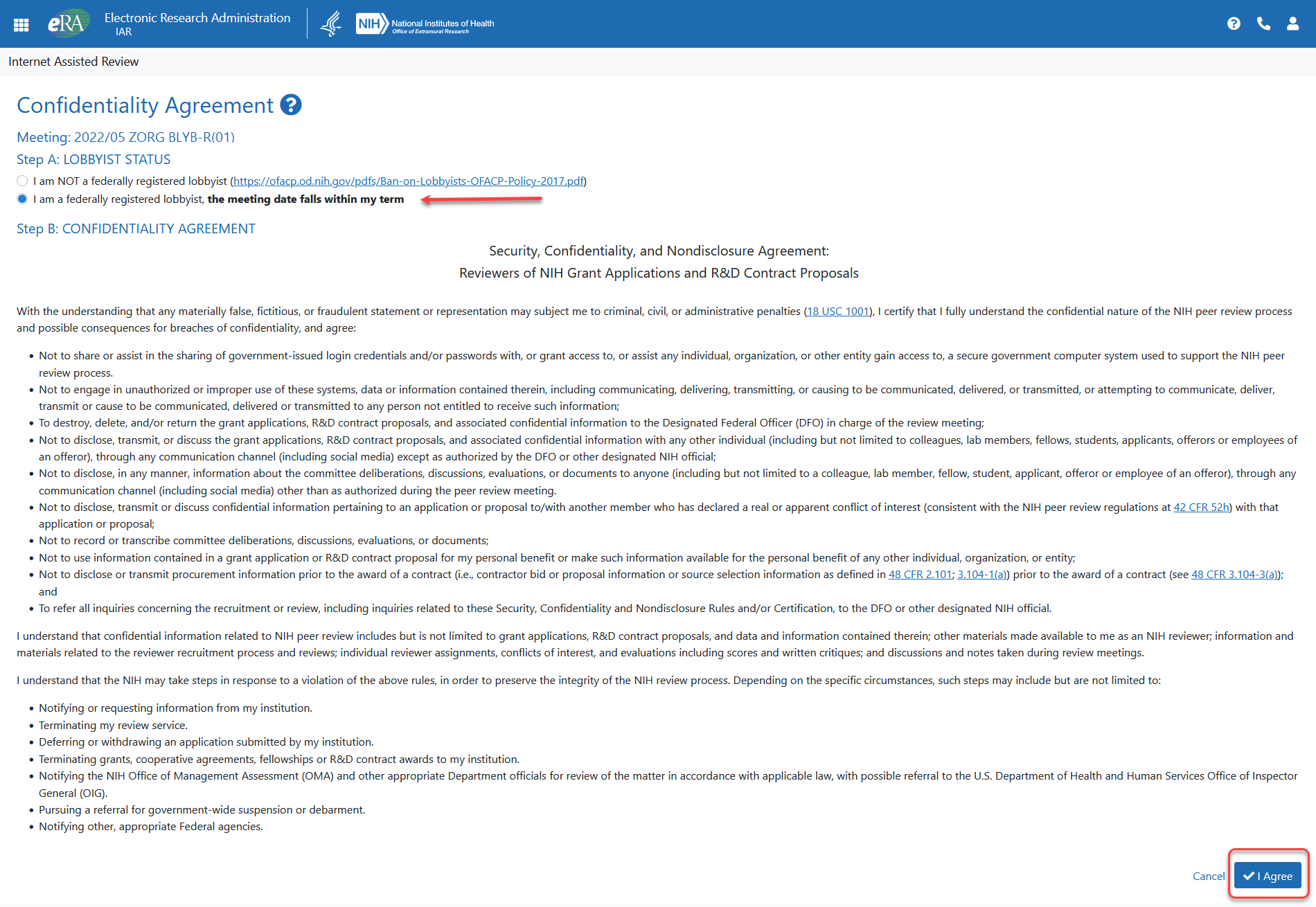
![]() The Confidentiality Agreement displays. This screen contains the NIH Confidentiality and Nondisclosure Rules. Please read the agreement carefully.
The Confidentiality Agreement displays. This screen contains the NIH Confidentiality and Nondisclosure Rules. Please read the agreement carefully.
-
Select the radio button indicating I am a federally registered lobbyist, the meeting date falls within my term.
-
Click the I agree button at the bottom of the screen.
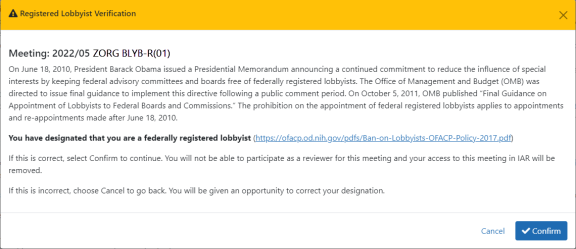
The Registered Lobbyist Verification screen displays. This screen shows that you have indicated that you are a federally registered lobbyist and explains the purpose for requesting the lobbyist status of reviewers. The screen provides a link for accessing additional information on this topic.
NOTE: Once you have indicated your status as a federally registered lobbyist, you will be denied access to the meeting. You will not have the ability to reverse the status yourself. If you mistakenly designate yourself as a federally registered lobbyist, you must contact the eRA Service Desk for assistance.
-
Only if you have accessed this screen and are NOT a federally registered lobbyist: Select the Cancel/Return button to return to the List of Meetings screen. Refer to Reviewer is Not a Federally Registered Lobbyist to continue.
-
Only if you want to continue to confirm your status as a federally registered lobbyist: Select the Confirm/Continue button.
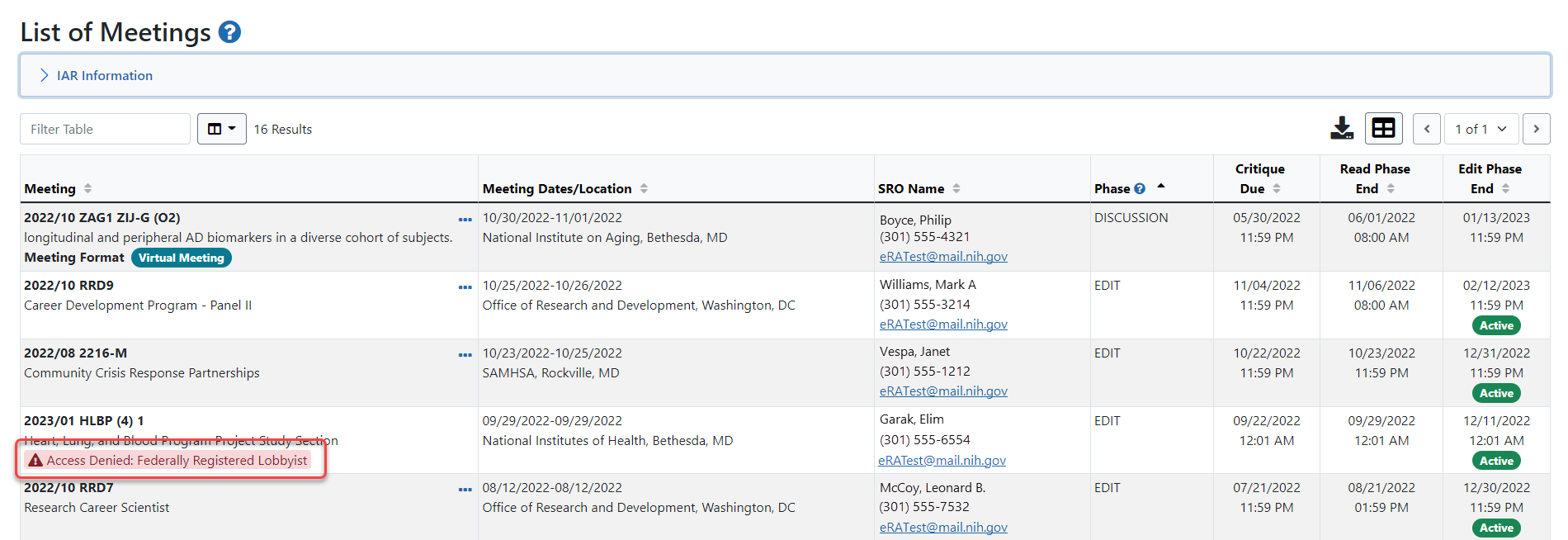
 The List of Meetings screen returns, displaying Access Denied: Federally Registered Lobbyist in the Meeting column. The SRO* receives an email whenever a potential reviewer designates himself as a federal lobbyist; however, it is a good idea to contact the SRO directly in this situation.
The List of Meetings screen returns, displaying Access Denied: Federally Registered Lobbyist in the Meeting column. The SRO* receives an email whenever a potential reviewer designates himself as a federal lobbyist; however, it is a good idea to contact the SRO directly in this situation.TIP: *Other Transaction Authority (OTA) — Some screens and terminology may differ to accommodate review of OTA, a type of award that is neither a grant nor a contract but a different way of funding that is used across NIH. These changes will typically not be visible to NIH or agency reviewers.